Unrivalled Lateral Flow Expertise for Developing High-Quality Assays
BBI Solutions has over 30-years of experience as an OEM partner for lateral flow assay development and manufacturings. Our highly skilled team has developed high-quality lateral flow assays for our global customers using our market-leading gold nanoparticle technology and lateral flow development materials. Whether you need end-to-end services or specific aspects of lateral flow assay development, we offer custom packages that can accommodate most applications or budgets.
Partner with BBI Solutions for Your Lateral Flow Assay Development Needs
Our unique combination of custom antibody development, lateral flow assay development and manufacturing services, and smartphone reader technology is shaping the future of lateral flow. We provide a one-stop-shop solution for all your lateral flow assay needs, including LFA development, manufacturing, antibody customisation, and gold particle supply.
Our collaborative approach to lateral flow development begins with a dedicated Business Development Manager and team of scientists to coordinate and deliver your custom Lateral flow project. The team also supports your operational organization to ensure that your assay meets your requirements.
The BBI Solutions Development Process
BBI Solutions offer a comprehensive step-by-step approach to assay development (detailed in the diagram below). For customers looking for complete customisable assay development, a typical programme will typically take 12 to 15 months (assay specific) and follows our robust development pathway with clear go/no go milestones between phases.
Our adaptive and communicative approach enables us to make quick decisions and change the project pathway to keep it on track and on time.
Typical LFA Project Phases
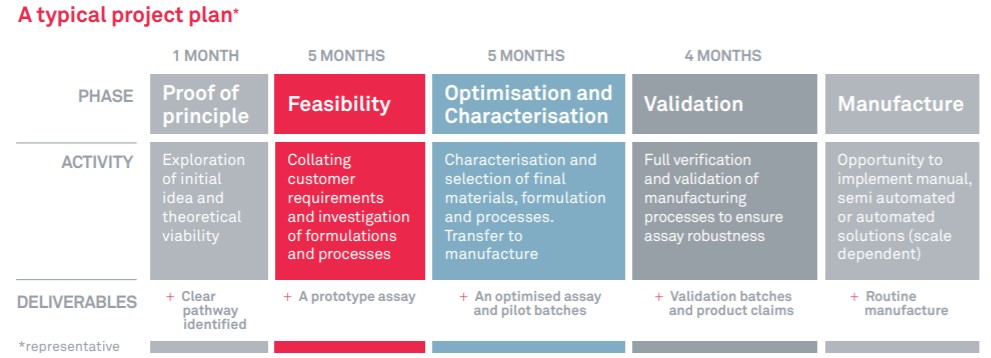
Our Crumlin site holds an FDA establishment registration and is certified to ISO13485:2016. We have experience working on projects with a range of different end-market regulatory requirements. Your dedicated team of scientists works exclusively on your assay to ensure rapid progress. At the end of the development cycle, we hand over the design history file so that you fully own your test.
A dedicated team of scientists work exclusively on your assay to ensure rapid progress. At the end of the development cycle, we will hand over the design history file, so that you fully own your test.
Experts in Assay Development
With over 250 successful projects to date and custom antibody solutions, BBI Solutions is the premier one-stop shop for lateral flow assay development.
- BBI Solutions is a market leader in gold nanoparticles and conjugates and have experience of working with a range of other labels such as latex, paramagnetic, and fluorescent nanoparticles.
- We utilise our own range of antibodies and antigens. We can also source commercially or use customer-specified antibodies/antigens.
- We have developed a large range of lateral flow assays, from fully quantitative reader-based assays to complex multiplex qualitative assays and simple two-line qualitative assays.
- We have assay development experience across a wide range of end-market applications, including Human Clinical, Pharmaceutical/Companion Diagnostics, Toxicology, Agri-diagnostics, Food Safety, Veterinary, and Defence markets.
- Within the human clinical IVD marketplace, we have experience working with multiple analytes, including Covid-19, HcG, LH, HIV, TB, Flu A and B, Calprotectin, Legionella, and Progesterone
- We have a wealth of reader experience from smart-phone technology, provided by our preffered partner Novarum DX, lab-based lateral flow readers from Axxin, Dialunox, IUL and even our customer's own reader designs.
BBI Solutions has the capacity to bring any lateral flow assay development project to life. Please contact our Customer Services Team directly to discuss your requirements on tel:+44 (0) 1495 363000 or alternatively you can submit an enquiry here.











